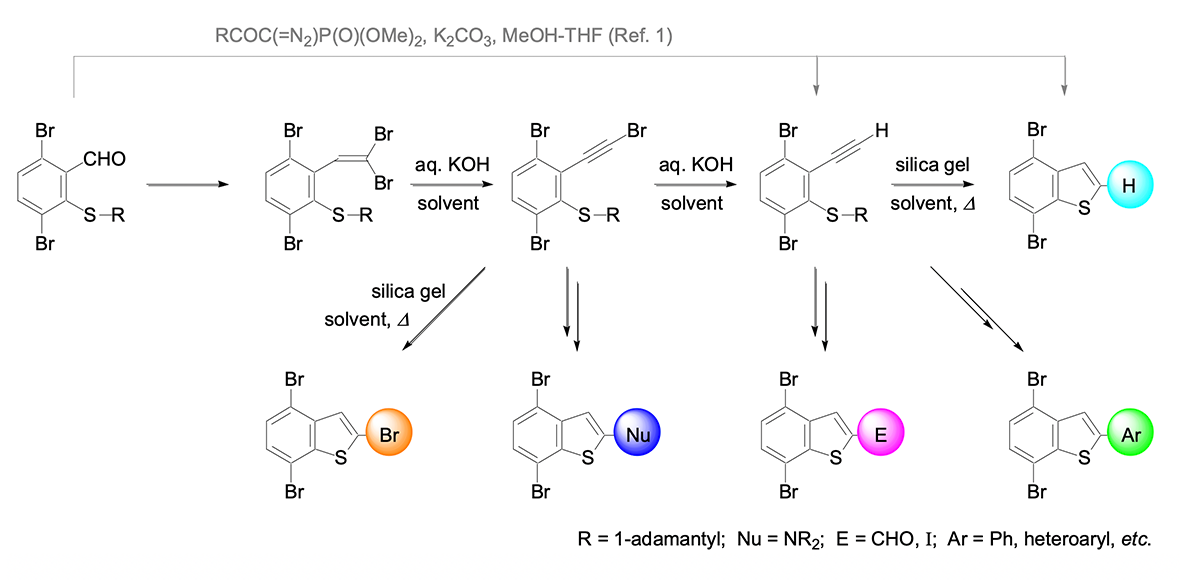
Mild Corey-Fuchs reaction conditions using DMSO – aq. KOH at room temperature were applied to preparation of 2-(1-adamantylsulfanyl)-1,4-dibromo-3-(ethynyl)benzene. Various substituents were introduced to the alkyne moiety of the (ethynyl)(sulfanyl)benzene or the intermediate (bromoethynyl)(sulfanyl)benzene, either by substitution reaction or by Sonogashira cross coupling. The alkynes thus obtained were converted to the corresponding 4,7-dibromobenzo[b]thiophene derivatives by the ‘silica gel-assisted cyclization’ method. Reactions of 4,7-dibromobenzo[b]thiophene and 2,4,7-tribromobenzo[b]thiophene were also investigated.
K. Toyota, S. Mikami, H. Tanaka, S. Yoshida, K. Iwai, K. Takahashi, H. Kishi, Y. Kikuchi, K. Saito, H. Yamaguchi, H. Mutoh
Heterocycles, 2020, 100(11), 1763.
DOI: 10.3987/COM-20-14302
https://www.heterocycles.jp/newlibrary/downloads/PDF/26923/100/11
(Ref. 1)
K. Toyota, H. Mutoh, H. Kishi, S. Mikami, H. Tanaka, S. Yoshida, D. Naganuma
Heterocycles, 2019, 98(10), 1355.
DOI: 10.3987/COM-19-14132
https://www.heterocycles.jp/newlibrary/downloads/PDF/26430/98/10
