A tungsten-silicon triple bonded complex reacted with two molecules of aldehydes at room temperature to give W-Si-O-C four-membered metallacycles via [2+2] cycloaddition and subsequent formyl hydrogen transfer from one aldehyde molecule to another. Upon gentle heating, the four-membered metallacycles underwent metathesis-like fragmentation cleanly to give W-C triple bonded complexes and “silanoic esters” having a Si=O bond, in a manner similar to that of metallacyclobutadiene, an intermediate of alkyne metathesis reactions, and dimerization of the latter products gave 1,3-cyclodisiloxanes.
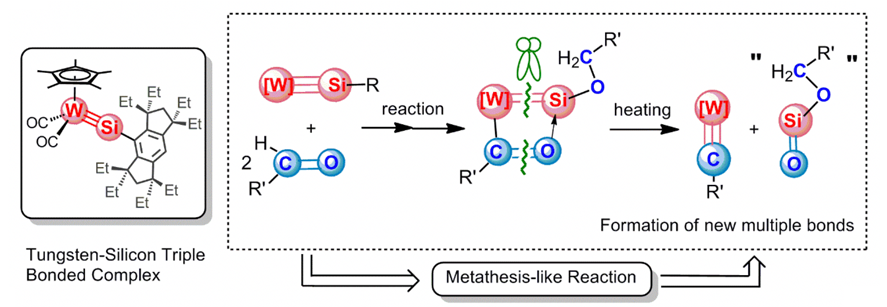
Reactions of a Silylyne Complex with Aldehydes: Formation of W-Si-O-C Four-Membered Metallacycles and Their Metathesis-Like Fragmentation
T. Yoshimoto, H. Hashimoto, N. Takagi, S. Sakaki, N. Hayakawa, T. Matsuo, H. Tobita,
Chem. Eur. J. 2019, 25, 3795-3798.
DOI: 10.1002/chem.201900457; https://doi.org/10.1002/chem.201900457
Recent Advances in the Chemistry of Transition Metal–Silicon/Germanium Triple-Bonded Complexes
H. Hashimoto, H. Tobita, Coord. Chem. Rev. 2018, 355, 362-379
DOI: 10.1016/j.ccr.2017.09.023; https://doi.org/10.1016/j.ccr.2017.09.023
