Metals are highly electrically conducting but have finite resistance. However, some metals show zero resistance state at sufficiently low temperature. The zero resistance is attributed to the superconducting state, which is useful for electric cables without power consumption, magnetic levitation trains, and electric power storages. However, the highest superconducting transition temperature has been still around 150 K (at ambient pressure) for a quarter century, hampering various applications. Only way to find room temperature superconductor is continuous efforts to explore novel superconductors.
Recently, a new compound Y2O2Bi whose crystal structure is the same as that of an iron-based superconductor was reported, but was not superconducting. However, this compound became superconducting by incorporating excess oxygens, in which the excess oxygens expand the distance between Bi square nets probably resulting in the superconducting transition. The superconducting phase showed the Berezinskii-Kosterlitz-Thouless transition due to the two dimensional Bi square net. By applying the chemical incorporation approach, it will be possible to make layered compounds superconducting.
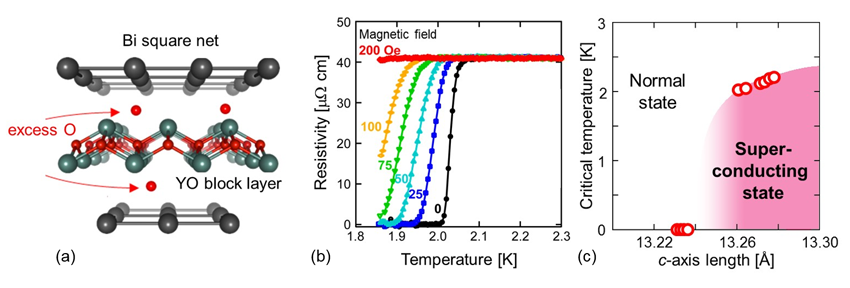
Figure. (a) Crystal structure of Y2O2Bi. Excess oxygens are thought to reside at interlayer space. (b) Temperature dependence of resistivity at different magnetic fields. (c) Relationship between superconducting transition temperature (critical temperature) and c-axis lattice constant. Excess oxygens increase the c-axis lattice constant, resulting in the emergence of superconductivity.
Ryosuke Sei, Suguru Kitani, Tomoteru Fukumura, Hitoshi Kawaji, Tetsuya Hasegawa
“Two Dimensional Superconductivity Emerged at Monatomic Bi2− Square Net in Layered Y2O2Bi via Oxygen Incorporation”
J. Am. Chem. Soc. 2016, 138, 11085−11088
DOI: 10.1021/jacs.6b05275
http://pubs.acs.org/doi/abs/10.1021/jacs.6b05275
