Nobuaki Kikkawa, Lingjian Wang, and Akihiro Morita have performed molecular dynamics simulations of ion transport through the interface between two immiscible electrolyte solutions—water and dichloromethane. Their approach reveals a hidden barrier to the passage of ions through the interface caused by the formation of a water finger that affects the rate of interfacial ion transfer (DOI: 10.1021/jacs.5b04375).
Experimental methods for studying the dynamics of liquid–liquid interfaces at a microscopic level are limited, and during the past two decades researchers have developed molecular dynamics simulations to study structural fluctuations at liquid–liquid interfaces associated with ion transfer. Using these simulations, Ilan Benjamin reported in 1993 the formation of a “water finger” by an ion passing through a water–oil interface (DOI: 10.1126/science.261.5128.1558). However, these approaches do not allow quantification of the “coordinate”, that is, a measure for the length, of the transient water finger trailed by the moving ion.
Using computations of the free energy surface during the passage of the ion from the water phase into the organic phase, the authors can now demonstrate the formation and breakage of a water finger. This finding explains why the observed rate of ion transfer differs from computations of this rate based on diffusion alone.
(Reprinted with permission from J. Am. Chem. Soc. 2015, 137, 8311. Copyright 2015 American Chemical Society)
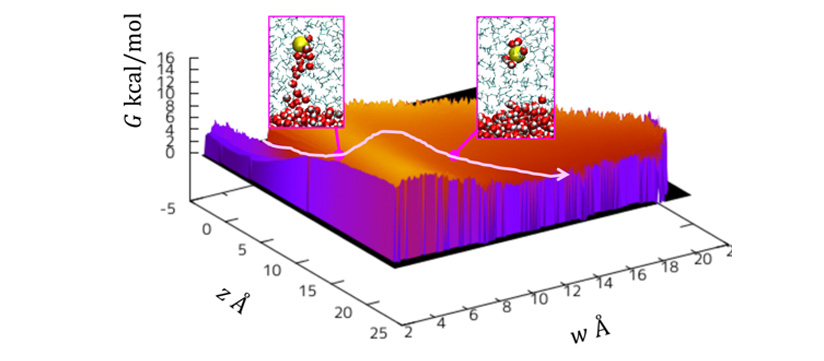
Figure: Free energy surface for the Cl- transport through water-oil interface. Left panel illustrates a structure with “water finger”, while the right panel with the broken water finger. The transition between the two states is associated with a barrier, which retards the transfer.
